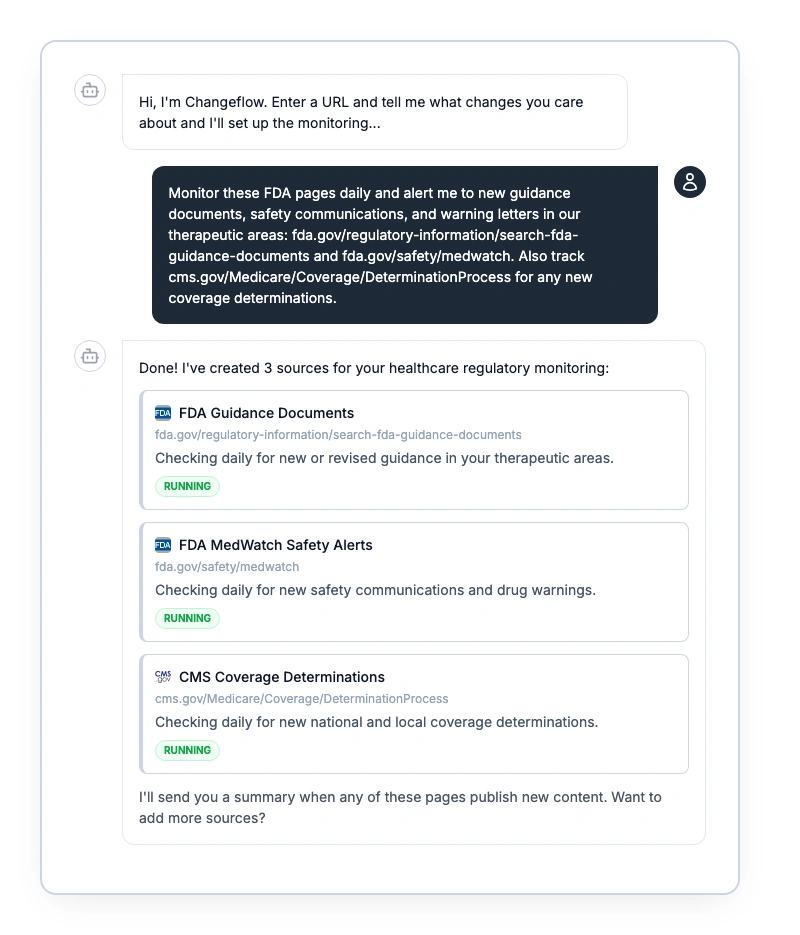
How pharma and healthcare compliance teams track FDA guidance changes, CMS policy updates, and Medicare coverage decisions. Build an automated monitoring workflow that catches changes before they cost you.
FDA and CMS Website Monitoring for Healthcare Compliance
The FDA quietly revises a guidance document on a Tuesday. It's posted to FDA.gov under a new URL path. No press release. No Federal Register notice. Your regulatory affairs team doesn't see it for six weeks.
By then, your competitor has already updated their labeling. Your QA team is still following the old version. And when the FDA inspector shows up, "we didn't know" is not an acceptable answer.
This is the daily reality for pharma RA teams, hospital compliance officers, and healthcare organizations that depend on FDA and CMS publications to stay compliant. The information is public. It's just scattered across hundreds of web pages that nobody has time to check manually.
This guide covers the specific FDA and CMS pages that matter, what changes to watch for, and how to build an automated monitoring workflow that catches updates before they become problems.
Why Healthcare Regulatory Monitoring Is Different
Healthcare is one of the most heavily regulated industries in the US. But the monitoring challenge isn't just volume. It's the structure of the information.
The FDA currently has 176 guidance documents under active development across all centers. CBER alone is planning 33 new guidances for 2026. CMS issued 432 transmittals in 2024, more than one per business day. State health departments add another layer of requirements that vary by jurisdiction.
Most of this content appears on agency websites before it reaches legal databases or industry newsletters. A NAVEX survey found that only 27% of chief compliance officers strongly agree they effectively monitor and track regulatory changes. In healthcare, where a missed guidance update can trigger a warning letter, that's a problem. Teams that adopt compliance monitoring software close this gap by automating detection across dozens of source pages.
Three things make healthcare monitoring uniquely difficult:
- Multiple agencies, overlapping jurisdiction. A pharmaceutical company might need to track CDER, CBER, ORA, CMS, DEA, FTC, and their state pharmacy boards. That's a dozen or more websites before you even start.
- Guidance vs. rules. The FDA publishes guidance documents that carry practical binding effect but aren't formal regulations. They don't always appear in the Federal Register. Legal research tools often miss them.
- Speed matters. When CMS changes a Medicare coverage determination, providers need to know immediately. Billing against an outdated policy means denied claims, payment clawbacks, and potential fraud liability.
Monitor regulatory pages automatically
Changeflow watches agency websites and tells you what changed and why it matters.
Free plan available. No credit card required.
FDA Pages Every Compliance Team Should Monitor
The FDA is organized into centers, each with their own web presence and publishing schedule. Here are the pages where changes appear first.
CDER (Drugs)
The Center for Drug Evaluation and Research handles everything related to prescription drugs, OTC drugs, and biological therapeutics.
Key pages to monitor:
- Guidance documents: Draft and final guidance on drug development, manufacturing, labeling, and post-market requirements
- New drug approvals: NDA and BLA approval letters, including labeling supplements
- Safety communications: Drug safety alerts, MedWatch notices, and REMS modifications
- Compliance programs: Compliance policy guides that define inspection priorities
CBER (Biologics)
The Center for Biologics Evaluation and Research covers vaccines, blood products, gene therapy, and cellular products.
Key pages:
- Guidance documents: Manufacturing, testing, and clinical requirements specific to biologics
- Licensure actions: BLA approvals and supplements
- Deviation reporting: Changes to reporting requirements for biological product deviations
CDRH (Medical Devices)
The Center for Devices and Radiological Health manages device approvals, recalls, and cybersecurity guidance.
Key pages:
- 510(k) decisions and PMA approvals: New device clearances and approval orders
- Device safety communications: Recalls, alerts, and field corrections
- Cybersecurity guidance: An increasingly active area, with new guidance on medical device cybersecurity published regularly since 2023
ORA (Inspections and Enforcement)
The Office of Regulatory Affairs is the FDA's enforcement arm.
Key pages:
- Warning letters: Published weekly, these signal enforcement priorities and interpretive positions
- Import alerts: Changes to import detention policies
- Inspection observations: Form 483 trends and common findings
Warning letters deserve special attention. They're not just enforcement actions against individual companies. They signal how the FDA interprets current regulations. When the FDA issues a cluster of warning letters on a specific topic, it usually means broader enforcement is coming.
CMS Pages for Healthcare Compliance
CMS manages Medicare, Medicaid, CHIP, and the Health Insurance Marketplace. For healthcare providers, CMS policy changes directly affect reimbursement, billing, and compliance obligations.
Medicare Coverage Determinations
CMS issues two types of coverage decisions that compliance teams need to track:
- National Coverage Determinations (NCDs): Apply nationwide. Published on the CMS Medicare Coverage Center. Changes here affect every Medicare provider offering the covered service.
- Local Coverage Determinations (LCDs): Issued by Medicare Administrative Contractors. Apply regionally. Published on the MAC's individual website. Miss a LCD change, and you're billing against an outdated coverage policy.
Billing and Coding Updates
CMS publishes changes to billing codes, payment rates, and claims processing rules through:
- Transmittals: Policy and system changes sent to MACs. Published on CMS.gov and often the first place where payment policy changes appear.
- MLN Matters articles: Medicare Learning Network publications that explain policy and system changes in plain language. These are the "translation layer" between formal policy and what your billing team needs to know.
- Fee schedule updates: Annual and mid-year changes to Medicare physician, outpatient, and inpatient payment rates.
State Medicaid Programs
If you operate across states, Medicaid monitoring gets complex fast. Each state runs its own Medicaid program within CMS guidelines. State plan amendments, waiver changes, and provider enrollment requirements all live on individual state agency websites.
No single database covers all 50 state Medicaid programs. Automated compliance monitoring of state agency pages is often the only way to catch these changes reliably. Law firms advising healthcare clients face the same multi-jurisdiction challenge when tracking obligations across federal and state agencies.
What Goes Wrong Without Monitoring
The consequences of missed FDA and CMS changes are concrete and expensive.
FDA Warning Letters
CDER warning letters jumped 50% in FY2025 compared to FY2024. Between July and December 2025, the FDA issued 327 warning letters, a 73% increase year over year. On September 9, 2025, the FDA issued over 60 warning letters in a single day targeting deceptive drug advertising.
Each warning letter cites specific regulatory violations, and many reference guidance documents that were updated months before the inspection. Companies that missed the update had no chance to come into compliance before the inspector arrived.
Common citation pattern: "The firm failed to follow current guidance on [topic]." Current being the key word. If you're still following guidance that was superseded by a newer version, that's a finding. See our guide on maintaining regulatory compliance for how to build processes that prevent this.
CMS Payment Clawbacks
When CMS changes a coverage determination or billing requirement, providers have a limited window to update their practices. Bill under the old rules after the effective date, and you're looking at claim denials, overpayment demands, and potential False Claims Act liability.
The American Hospital Association reports that the average community hospital spends $7.6 million per year on regulatory compliance, with 59 FTEs dedicated to it. A single denied Medicare claim isn't a crisis. But when a coverage policy changes and your billing team doesn't know about it for three months, you're looking at hundreds or thousands of improperly billed claims. The math gets ugly fast.
Clinical Trial Delays
For pharma companies running clinical trials, an FDA guidance change can alter protocol requirements, consent form language, or endpoint definitions. If your team doesn't catch the updated guidance before your next FDA meeting, you risk protocol amendments, delayed reviews, and extended timelines.
The cost of a single day's delay in a Phase III clinical trial can exceed $600,000 in lost revenue for a blockbuster drug. Missing a guidance update that triggers a protocol amendment can add months.
HIPAA Enforcement
The Office for Civil Rights (OCR) enforces HIPAA and publishes guidance on its website about privacy, security, and breach notification requirements. OCR has ramped up enforcement significantly, with penalties reaching $4.75 million for individual violations.
Guidance changes on topics like telehealth privacy, patient access rights, and security rule requirements appear on the HHS OCR website before they reach industry publications.
How to Build an FDA/CMS Monitoring Workflow
Manual monitoring doesn't scale. Here's a realistic workflow that actually works.
Step 1: Map Your Source Pages
Start with a source inventory. For a mid-size pharmaceutical company, the list typically looks something like this:
FDA sources (15-25 pages):
- CDER guidance documents
- CBER guidance documents (if applicable)
- Warning letters
- Safety communications
- Drug approval letters and labeling supplements
- Compliance policy guides
- Federal Register FDA entries
CMS sources (10-20 pages):
- National Coverage Determinations
- Local Coverage Determinations (your region's MAC)
- Transmittals
- MLN Matters articles
- Fee schedule updates
- Proposed and final rules
Other federal (5-10 pages) (see our broader guide on monitoring government websites for the full picture):
- DEA scheduling actions
- FTC health claims enforcement
- OCR HIPAA guidance
- OIG advisory opinions
That's 30-55 pages minimum. Nobody is checking all of these manually every day.
Step 2: Set Up Automated Monitoring
Use a website monitoring tool to track changes on each source page automatically. The tool checks the page on a schedule (typically every few hours), detects when content changes, and sends an alert.
Here's what to look for in a monitoring tool for FDA compliance monitoring:
- AI filtering: FDA.gov pages change frequently. Menu updates, footer changes, layout tweaks. You need AI that reads the page content and tells you whether the change is substantive or cosmetic.
- Link following: FDA guidance documents are often listed on index pages. The tool should follow links to individual guidance PDFs and alert you when new documents appear.
- Custom briefs: Tell the tool what you care about. "Alert me to changes in CDER guidance related to post-market safety reporting" is more useful than "alert me to any change on this page."
Changeflow handles all three. You give it a URL and a brief description of what you care about. The AI watches the page, filters out noise, and sends you a summary when something relevant changes.
Step 3: Route Alerts to the Right People
Not every FDA change affects every team. Route alerts based on source and content:
- CDER guidance changes → Regulatory affairs
- Warning letters in your product area → Quality and compliance
- CMS coverage determinations → Medical affairs and billing
- Safety communications → Pharmacovigilance
- HIPAA guidance → Privacy officer
Most monitoring tools support email alerts, Slack notifications, and webhook integrations that can feed into your existing workflow tools.
Step 4: Review and Assess
When an alert arrives, someone needs to read it and decide: does this affect us?
Good regulatory change management separates detection from assessment. The monitoring tool handles detection. Your subject matter experts handle assessment. Don't make your SMEs also responsible for checking websites. That's a waste of their time and expertise.
FDA vs. Legal Research Databases
A common question: "We already have Westlaw/LexisNexis. Don't they cover FDA?"
Partly. Legal research platforms index statutes, case law, and Federal Register publications. They're good at what they do.
But they don't systematically cover:
- Draft guidance documents that haven't been published in the Federal Register
- FDA FAQ pages that are updated without formal notice
- Compliance policy guides that change how inspectors evaluate your facility
- Warning letter trends that signal upcoming enforcement priorities
- CMS transmittals that modify billing requirements
- State Medicaid program changes published on individual state websites
This is the "regulatory dark matter" problem. The changes that carry practical binding effect but don't appear in formal legal databases. Our regulatory compliance examples guide shows specific cases where teams caught these changes through source monitoring.
Enterprise regulatory intelligence platforms like FiscalNote and Thomson Reuters Regulatory Intelligence offer broader coverage than legal research tools. But they start at $50,000+ per year. For the awareness layer specifically, a source monitoring tool like Changeflow covers the same FDA and CMS pages at a fraction of the cost.
Getting Started
If you're building healthcare compliance monitoring from scratch, start small.
Pick your three highest-risk FDA source pages. Set up monitoring. See what changes come in over the first two weeks. You'll quickly learn which pages update frequently and which are quiet, which changes matter and which are noise.
Then expand. Add CMS sources. Add your state pharmacy and medical boards. Build out the source inventory over time. Subscribe to FDA GovDelivery and CMS email alerts as a free baseline layer, and add automated page monitoring for the guidance and FAQ pages those email lists don't reliably cover.
The organizations that catch FDA guidance changes first have a real compliance advantage. Not because they're smarter, but because they set up a system that doesn't depend on someone remembering to check a bookmark.
That's the whole point. Build the awareness layer right, and the rest of your compliance program works better.
Track FDA and CMS changes automatically
Get alerts when FDA guidance, CMS policies, or Medicare coverage decisions change. AI reads the page and tells you what matters.
Try Changeflow FreeNo credit card required
More from Learn

Wayback Machine for Legal Research: A Law Librarian's Guide
Half the URLs in old briefs are dead. Here is how law librarians use the Wayback Machine and stronger archives to verify citations, build timelines, and support litigation.

Law Librarian Current Awareness Audit Checklist (2026)
A 27-question audit covering coverage, cadence, relevance, distribution, resilience, and value. Built for law librarians and knowledge lawyers running current awareness at Am Law 200 firms.
Audit-Ready Web Archives: SEC 17a-4, FDA, and HIPAA Rules
Free archive tools fail under regulatory audit. Here is what SEC 17a-4, FDA 21 CFR Part 11, and HIPAA require from a web archive that holds up under examination.