How compliance teams detect, assess, and act on regulatory changes. Real enforcement data, the dark matter problem, and how to build an automated monitoring workflow.
A rule changes on a Friday afternoon. A guidance document is quietly revised. A state agency updates an FAQ page that nobody thought to check.
Three months later, your organization gets fined because it was following the old version. That's not hypothetical. It happens constantly. And it's the core problem regulatory change management exists to solve.
This guide covers how compliance teams actually manage regulatory change, not the textbook version. We'll look at the enforcement data, the sources most teams miss, and how to build an awareness layer that doesn't depend on someone remembering to check 50 bookmarks every morning.
What Is Regulatory Change Management?
Regulatory change management is the process of detecting, assessing, and responding to changes in laws, regulations, and agency guidance that affect your organization.
Simple definition. The execution is where teams struggle.
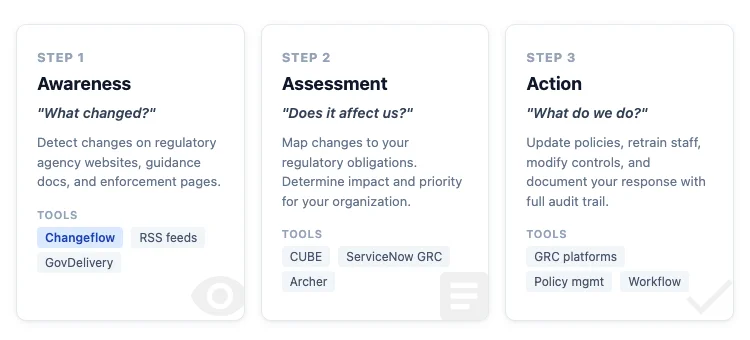
The workflow breaks into three steps:
- Awareness: What changed? A new rule, a revised guidance, an updated FAQ, an enforcement action that signals a new interpretation.
- Assessment: Does it affect us? Map the change against your regulatory obligations and determine impact.
- Action: What do we do? Update policies, train staff, modify processes, file required reports.
Most compliance failures happen at step one. Not because the team is incompetent, but because the sources are scattered across dozens of agency websites, and nobody has a reliable system for catching every update.
A compliance officer at a mid-size bank might need to monitor the SEC, CFPB, OCC, FinCEN, OFAC, their state banking regulator, and FINRA. That's seven agencies minimum, each with their own website structure and publishing cadence. Tracking government website changes across all of them manually is a losing battle. Miss one update on one page, and you have a gap.
Monitor regulatory pages automatically
Changeflow watches agency websites and tells you what changed and why it matters.
Free plan available. No credit card required.
Why It Matters More Than Ever
The enforcement numbers tell the story.
The SEC collected $8.2 billion in penalties and disgorgement in FY2024, their highest total ever. GDPR fines have exceeded EUR 5.88 billion since the regulation took effect. TD Bank paid $3.09 billion in AML penalties in 2024, the largest Bank Secrecy Act enforcement action in history.
And fines are accelerating. Regulatory enforcement penalties rose 417% in the first half of 2025 compared to the same period the year before.
The gap between what regulators expect and what teams actually track is growing. A Thomson Reuters survey found that 76% of compliance teams still manually scan for regulatory changes. They're checking websites, reading newsletters, and asking colleagues if they've seen anything new.
That worked when regulations changed slowly. It does not work anymore.
The pace of regulatory change has increased dramatically. The Federal Register published over 90,000 pages of rules and proposed rules in 2024. And that's just the formal rulemaking. It doesn't include the thousands of guidance documents, bulletins, FAQ updates, and enforcement actions published on individual agency websites every year.
The Regulatory Dark Matter Problem
Here's something the GRC vendor brochures won't tell you: the biggest compliance risks aren't in the Federal Register. They're in guidance documents, FAQ pages, and staff bulletins that most monitoring systems completely miss.
How big is this problem? When the Trump administration issued Executive Order 13891 in 2019, requiring agencies to catalog their guidance documents, federal agencies found over 73,000 guidance documents across the government. These are documents that carry practical binding effect on regulated entities but were never published through the formal rulemaking process.
Legal research platforms like Westlaw and LexisNexis index statutes and case law. They're good at what they do. But they don't systematically cover agency guidance, FAQ pages, or the informal publications where many actionable changes first appear.
We call this "regulatory dark matter." The changes that affect your compliance posture but don't show up in the systems most teams rely on.
Some real examples of regulatory compliance failures from this gap:
- The FDA updates a guidance document on drug labeling. It's posted on FDA.gov but never published in the Federal Register. Your labeling team doesn't see it for three months.
- A state attorney general updates their FAQ on privacy law enforcement. The change clarifies what "reasonable security" means for your industry. You find out about it from an enforcement action against your competitor.
- OFAC adds names to the SDN list on a Friday. Your compliance screening system uses a weekly data feed. For five days, you're processing transactions involving sanctioned entities.
These aren't edge cases. They're the normal experience of compliance teams that rely on formal publications and email newsletters for their regulatory awareness.
The 3-Step Regulatory Change Workflow
Let's break down each step and the tools that handle it.
Step 1: Awareness (What Changed?)
This is the detection layer. Something changed on a regulatory website, and you need to know about it.
What tools handle this: Source monitoring platforms like Changeflow, website change detection tools, RSS feeds, and government email subscriptions.
What good looks like: Every regulatory source that matters to your organization is monitored automatically. When a page changes, you get an alert within hours, not days. AI filters out navigation changes and menu updates, surfacing only substantive content changes.
What most teams actually do: Check bookmarks manually. Subscribe to a few email newsletters. Ask colleagues. Hope.
Step 2: Assessment (Does It Affect Us?)
Once you know something changed, you need to determine whether it affects your regulatory obligations.
What tools handle this: GRC platforms like CUBE, ServiceNow GRC, and Archer. These map regulatory changes to your obligation register and flag items that require review.
What good looks like: A change triggers an automated workflow that routes it to the right subject matter expert. The SME reviews, determines impact, and either dismisses it or initiates an action plan.
What most teams actually do: Forward emails. Have meetings. Track in spreadsheets.
Step 3: Action (What Do We Do?)
The change affects you. Now you need to update policies, retrain staff, modify controls, or file reports.
What tools handle this: Workflow and project management tools, GRC platforms, policy management systems.
What good looks like: Clear ownership, deadlines, and audit trails. Regulators can see exactly when you became aware of a change and what steps you took to respond.
Here's what matters: most teams invest heavily in steps 2 and 3 (GRC platforms, obligation mapping, workflow tools) while leaving step 1 barely covered. They're building elaborate assessment frameworks on top of a broken awareness layer.
If you don't catch the change, the best obligation mapping tool in the world won't help you.
What to Monitor (And Where to Find It)
The specific sources depend on your industry, but here's a starting framework.
Federal Agencies
Every regulated entity should be monitoring their primary federal regulators. The actual pages where changes appear first:
- FDA: Guidance documents page, warning letters, safety communications, drug approval letters. See our full FDA and CMS monitoring guide.
- SEC: Staff bulletins, no-action letters, enforcement releases, proposed and final rules. See our full SEC EDGAR monitoring guide.
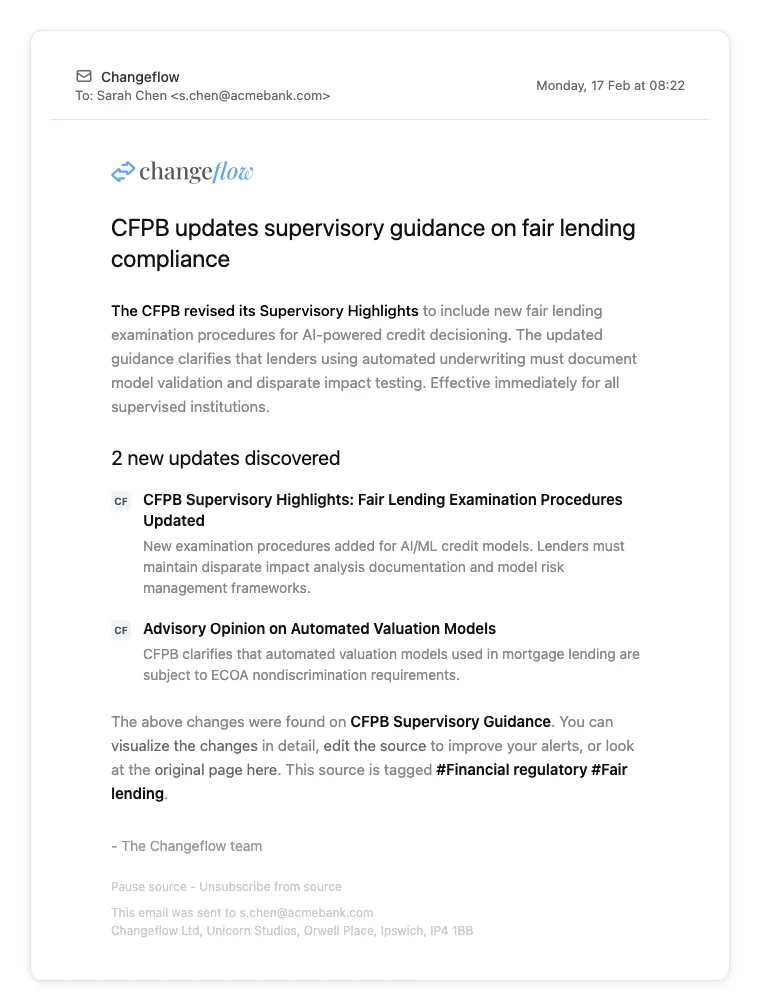
- CFPB: Supervisory guidance, enforcement actions, advisory opinions
- FinCEN: AML advisories, geographic targeting orders, beneficial ownership guidance
- OFAC: SDN list updates, general licenses, compliance guidance
For FDA compliance monitoring and SEC filing monitoring specifically, Changeflow has pre-built source templates that cover the key pages.
State Agencies
State-level monitoring is where most teams fall short. There are 50 state attorneys general, 50 insurance commissioners, 50 banking departments, and dozens of other state regulators with their own websites and publishing schedules.
If you operate nationally, you can't realistically monitor every state manually. Automated page monitoring is how compliance teams cover the breadth without hiring a state regulator analyst per jurisdiction. Focus your manual review on your highest-risk states and let automation handle the rest.
International Regulators
For organizations with global operations:
- UK FCA: Handbook updates, policy statements, consultation papers
- EU/ESMA: Technical standards, Q&A updates, opinions
- EDPB: Guidelines, opinions, consistency decisions (for GDPR)
The challenge with international monitoring is language and jurisdiction. A single regulatory change might require different responses in different countries.
Industry Self-Regulatory Bodies
Don't forget FINRA, PCAOB, stock exchange rules, bar association ethics updates, and industry standards bodies. These aren't government agencies, but their rules carry enforcement weight.
Manual vs Automated Monitoring
The Manual Approach
Most compliance teams start here. Someone opens 20-50 bookmarks every morning, scans for changes, and flags anything relevant. It's the same problem law librarians face with current awareness, and it breaks at the same point: one person can't cover the agency-website surface area.
The problems are obvious:
- It doesn't scale. One person can realistically check 30-40 pages per day. Most organizations need to monitor hundreds.
- People miss things. Especially subtle changes. An edited paragraph in a 200-page guidance document? Good luck catching that manually.
- No audit trail. When did you become aware of a change? You checked the website on Tuesday, but did you actually read the updated section? Can you prove it?
- Newsletter lag. Government email subscriptions are often delayed by days or weeks. And they don't cover everything. The FDA's GovDelivery system is better than most, but it still misses guidance document revisions.
- Staff turnover kills you. When your one compliance analyst who checks websites every morning leaves, the whole system breaks.
The Automated Approach
Automated monitoring tools track changes on regulatory pages and alert you when content changes. The better ones use AI to filter noise from signal.
Here's why the AI part matters: regulatory pages change constantly for reasons that don't matter. Navigation updates, footer changes, cookie banner modifications, server timestamps. Without filtering, you get hundreds of false alerts that train your team to ignore notifications.
With AI filtering, the tool reads the page, understands what changed, and tells you whether it's a substantive content change or just website maintenance. You get the updates that matter, not the noise.
A compliance monitoring setup through Changeflow works like this:
- Add the regulatory pages you need to track (or use pre-built source templates for common agencies)
- Tell the AI what you care about, like "guidance updates affecting drug labeling" or "enforcement actions against financial institutions"
- Get daily or real-time alerts when relevant changes happen
- AI summarizes what changed and why it matters
One dashboard. All your regulatory sources. No more morning bookmark ritual.
Regulatory Change Management Software Compared
The market breaks into distinct layers. Understanding which layer you need determines which tool fits.
| Tool | Focus | Best For | Typical Cost |
|---|---|---|---|
| Changeflow | Source monitoring + AI filtering | Awareness layer. Detecting changes on agency websites | From $99/mo |
| CUBE | Obligation mapping + regulatory intelligence | Assessment layer. Mapping changes to obligations | $50-150K/yr |
| FiscalNote | Legislative tracking + political intelligence | Tracking bills and proposed legislation | $50K+/yr |
| Thomson Reuters Regulatory Intelligence | Regulatory news + content | Research and alerting on regulatory developments | $50-200K/yr |
| ServiceNow GRC | GRC workflow + compliance management | Action layer. Managing compliance programs end-to-end | $100K+/yr |
| Archer (RSA) | Risk and compliance management | Enterprise risk management with regulatory workflow | $100K+/yr |
Let's be honest about positioning. Changeflow handles the awareness layer: monitoring sources, detecting changes, and filtering noise. We don't do obligation mapping or GRC workflow management. CUBE and FiscalNote don't monitor individual agency web pages for changes.
For most compliance teams, the right answer is a combination. Use an affordable monitoring tool like Changeflow for the awareness layer. Use a GRC platform for assessment and action. The enterprise platforms are good at what they do, but they're expensive, and they don't replace the need for source monitoring.
If you're evaluating alternatives, see our detailed comparisons of Changeflow vs Visualping, Changeflow vs FiscalNote, and Changeflow vs Thomson Reuters for the monitoring layer specifically.
What About Feedly, Meltwater, and Media Monitoring?
These are different categories. Feedly aggregates RSS feeds. Meltwater monitors news coverage. Neither monitors the actual regulatory source pages where changes appear first.
By the time a regulatory change shows up in a news article or RSS feed, it's been public for hours or days. For compliance purposes, you want the primary source, not secondary coverage.
Building Your Regulatory Change Management Framework
Here's a practical framework you can implement this quarter. No enterprise sales cycle required.
1. Inventory Your Regulatory Obligations
List every regulation, standard, and guideline that applies to your organization. Group by:
- Agency: FDA, SEC, state AG, etc.
- Type: Statute, regulation, guidance, enforcement precedent
- Priority: Critical (directly affects operations) vs. important (affects peripherally) vs. watch list (could affect in future)
You probably already have most of this in a spreadsheet or GRC system. If not, start with your top 10 regulatory risks.
2. Map Sources to Obligations
For each obligation, identify the primary source pages where changes would appear. This is the step most teams skip. They know they need to track FDA guidance, but they haven't identified the specific URLs.
Build a source map:
- FDA Drug Guidance Documents:
fda.gov/regulatory-information/search-fda-guidance-documents - SEC Staff Bulletins:
sec.gov/corpfin/staff-bulletins - Your state AG Privacy Page:
[state].gov/privacy-enforcement
3. Set Up Automated Monitoring
Add your source URLs to a monitoring tool. For regulatory intelligence specifically, set monitoring frequency based on risk:
- Critical sources: Check every 6-12 hours (enforcement pages, primary regulator)
- Important sources: Check daily (secondary regulators, industry bodies)
- Watch list: Check weekly (emerging regulations, other jurisdictions)
Configure AI filtering to match your brief. "FDA guidance changes affecting pharmaceutical labeling" is more useful than "any change on FDA.gov."
4. Define Your Escalation Workflow
When the tool detects a change:
- Triage: Is this a substantive regulatory change or website maintenance?
- Route: Send to the right subject matter expert based on topic and jurisdiction
- Assess: Determine impact on existing obligations within 48 hours
- Act: If action required, assign tasks with deadlines
- Document: Create an audit trail of awareness, assessment, and response
5. Review and Improve
Quarterly, review your source list. Are there agencies you should be monitoring that you aren't? Have any of your regulatory risks shifted? Did you miss any changes that you should have caught?
The framework doesn't need to be perfect on day one. Start with your highest-risk sources and expand from there.
For teams getting started with regulatory monitoring, Changeflow offers horizon scanning templates for common regulatory frameworks. Add your agency URLs, configure your brief, and start getting alerts. From $99/month, no enterprise contract required.
Monitor regulatory pages automatically
Get alerts when FDA, SEC, or state agency pages change. AI filters the noise and tells you what matters.
Try Changeflow FreeNo credit card required
More from Learn

Wayback Machine for Legal Research: A Law Librarian's Guide
Half the URLs in old briefs are dead. Here is how law librarians use the Wayback Machine and stronger archives to verify citations, build timelines, and support litigation.

Law Librarian Current Awareness Audit Checklist (2026)
A 27-question audit covering coverage, cadence, relevance, distribution, resilience, and value. Built for law librarians and knowledge lawyers running current awareness at Am Law 200 firms.
Audit-Ready Web Archives: SEC 17a-4, FDA, and HIPAA Rules
Free archive tools fail under regulatory audit. Here is what SEC 17a-4, FDA 21 CFR Part 11, and HIPAA require from a web archive that holds up under examination.